Introducing Endless Mode: A New Games & Anime Site from Paste
The U.S Food and Drug Administration has just approved Dupixent, a new treatment option for people with moderate-to-severe eczema.
Dupixent works as an injection under the skin, and was developed for patients whose condition can not be controlled by topical treatments. However, it also gives patients who would typically use a topical treatment a break from applying creams to the skin multiple times per day.
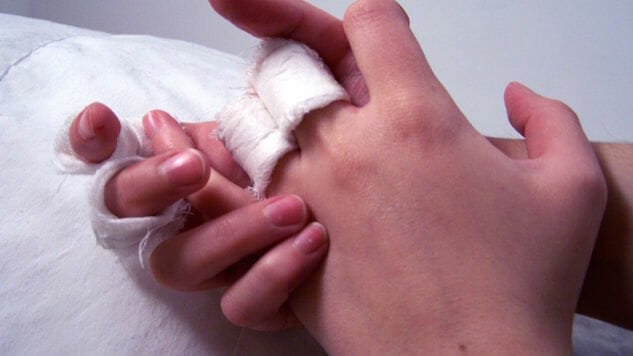
Eczema is a condition when patches of skin become inflamed, itchy, red, cracked and rough. Most people outgrow the condition as children, but some continue to endure symptoms into adulthood. The causes of eczema are unknown, however it is believed to develop due to both genetic factors such as having parents with atopic diseases and environmental factors such as irritants, allergens, microbes, extreme temperatures, food, stress and hormones.
Dupixent was approved by the FDA after going through 3 clinical trials that included roughly 2,100 adults with moderate-to-severe eczema, whose symptoms could not be controlled by topical treatments. After 16 weeks, those who received Dupixent had clearer skin and less itching than those who received the placebo.
The most common side effects of the drug included injection site reactions, cold sores in the mouth and lips, and eye/eyelid inflammation. The FDA recommended those experiencing eye symptoms to see a doctor.
Dr. Julie Beitz, director of the Office of Drug Evaluation III in the FDA’s Center for Drug Evaluation and Research, said in a news release that “eczema can cause significant skin irritation and discomfort for patients, so it is important to have a variety of treatment options available to patients, including those patients whose disease is not controlled by topical therapies.”
Photo: Vanina W., CC-BY
Elizabeth Chambers is a health intern with Paste and a freelance writer based out of Athens, Georgia.